SHOEBOX PureTest is the world’s first automated iPad-based audiometer. More than simply portable, SHOEBOX PureTest performs diagnostic hearing testing using a modified Hughson-Westlake protocol. The system is FDA and Health Canada listed, conforms to current ANSI S3.6 requirements, and produces the same clinically valid frequency thresholds as conventional audiometers. SHOEBOX is:
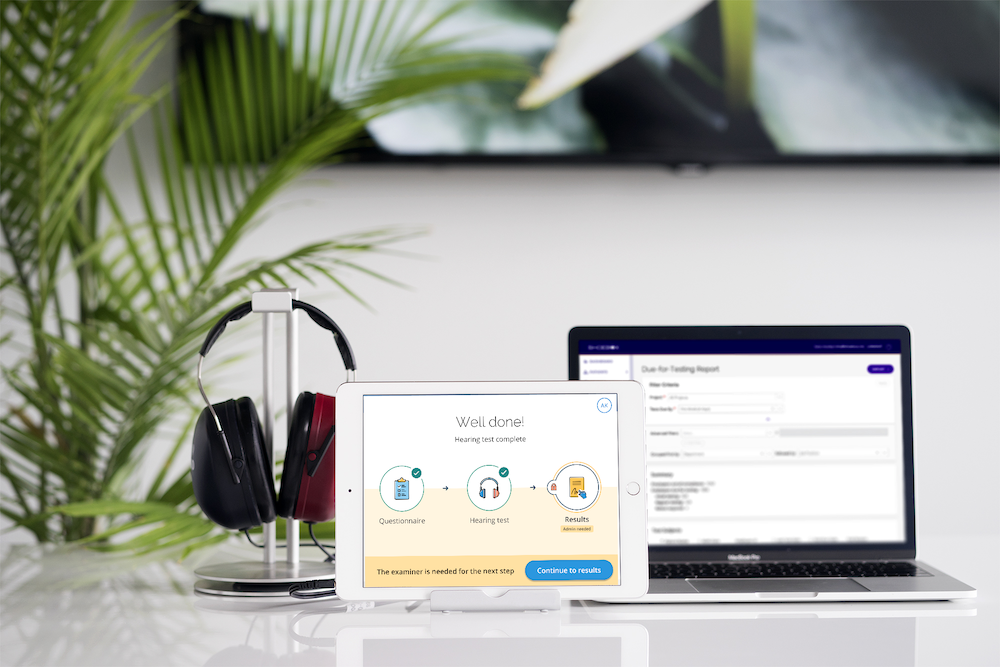
SHOEBOX Consult is a software-based digital audiometer designed to help deliver more patient-centric hearing services. It enables hearing care professionals to perform diagnostic evaluations while delivering an innovative and optimal patient experience.
SHOEBOX Consult is:
• Optimized for use outside of a sound booth
• A software-based, portable audiometer
• Highly intuitive, with a user-friendly interface
• Available with an optional teleaudiology add-on to enable remote diagnostic evaluations including video otoscopy
AD226 can be used as a fully self-contained, standalone audiometer or can be use with a computer to offer all the benefits of a PC-based audiometer such as full integration with Noah or OtoAccess software systems, electronic health records, or hospital information systems.
The AD 226 powerful and portable (it weighs less than 3 pounds). It allows you to perform air and bone conduction audiometry with pure tone, warble and pulsed tones, and narrowband noise and white noise masking. AD 226 includes a built-in talk forward microphone to enable communicating with the patient in the sound booth.

The FONIX 8000 is the world’s most advanced hearing aid test system, featuring the unique Polar Plot Sound Chamber, capable of performing automatic polar plot measurements of directional hearing aids. Although the results are not a replacement for laboratory testing using an anechoic chamber, the 8120 Polar Plot chamber provides the hearing health professional with a clinical tool for verifying approximate directionality characteristics of the hearing aid. As directional hearing aids continue to grow in popularity, this feature has become essential in the modern hearing aid clinic.

Cortical Auditory Evoked Potential (CAEP) measurements are used to detect whether particular sounds produce an electrical response in a patient’s brain. HEARLab provides the hearing health professional with an easy-to-use method of performing this measurement.
A transducer such as an insert earphone, bone vibrator, or sound field speaker is used to deliver the test signal to the patient who is passively alert. Electrodes placed on the patient’s head measure the cortical response. A statistical analysis of each response (a “p-value”) is automatically calculated to determine the probability that a CAEP signal was present. The clinician can verify this analysis with a visual examination of the measured cortical response.
CAEP can be tested on patients who are unable or unwilling to communicate to the clinician about whether they can hear a signal. This could include infants and small children who have not yet developed language skills, and adults who are...
